According to WHO, breast cancer is the most common cancer among women, impacting 2.1 million women globally each year. In India, a woman is diagnosed with breast cancer every four minutes and its incidence is on the rise in both rural and urban India. Early detection is recognized as the best way to improve breast cancer outcomes and survival rates.
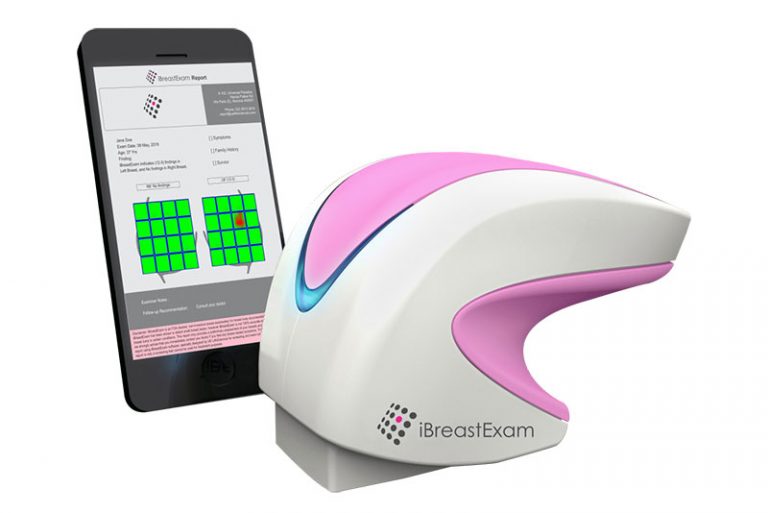
In 2007, the year the founder Mihir Shah was getting married, his soon-to-be mother in law was diagnosed with breast cancer. She underwent chemotherapy and survived, wearing a wig to the wedding. The incident spurred Mihir to establish UE Lifesciences with co-founders Matthew Campisi and Bhaumik Sanghvi. When they started to dig deeper, they learnt that over 90% of women in the developing world did not have access to any mechanism of early detection of breast cancer. To fill this gap, they came up with ‘iBreastExam’, an early screening device for breast cancer.
iBreastExam uses innovative sensor and material technology combined with software computing to accurately identify cancerous lesions and tumours. This breakthrough technology has the potential to dramatically improve the accessibility and affordability of breast cancer screening. It is a portable, radiation-free and non-invasive US FDA approved device ensuring that the screenings are safe, pain-free and private. They have also proposed an innovative pay-per-use model – instead of targeting direct sales – which allows doctors in every corner of the country to start screening women for breast cancer at the earliest.
The 2nd generation iBreastExam sensor technology has improvements (spatial resolution, data frequency, power consumption, ease of use, clinical efficacy) by orders of magnitude over the first generation technology, helping expand access to many more women globally.
Today, iBreastExam has reached over 350,000 women in 12 countries and has aided in the early diagnosis of 150 cases of breast cancer. With devices currently on the market, over 1 million women will soon receive a safe, affordable and radiation-free breast exam in the convenience of a community health center or their home. iBreastExam is commercially market cleared by US FDA, CE mark and eight other regulatory bodies in Southeast Asia, Latin America and Middle East.
In 2017, GE Healthcare announced a distribution partnership with UE LifeSciences to bring iBreastExam to women in 25 countries. UE LifeSciences has also forged strategic partnerships with big pharma and leading medical device distribution channels. The Pfizer Foundation and the Bayer Cares Foundation are currently sponsoring screening programs in Burma and Brazil.

Mihir is Founder and CEO of UE LifeSciences and a computer engineer by training. He has 15+ years of experience in bringing disruptive science from academia to the clinic.

FORMER CHAIRPERSON, THERMAX
Ms. Anu Aga is the former Chairpersonof one of India’s leading business houses –Thermax India. Since October 2004, she is serving as Director on the Thermax Board. After retiring from Thermax she took to social workand in 2010 was awarded the Padma Shri by the Government of India.She is also the Chairperson of Teach For India.She was nominated to Rajya Sabha, the Upper House of Indian Parliament by the Indian presidentin 2012. Earlier, she was a member of the National Advisory Council (NAC) chaired by Ms. Sonia Gandhi.

CHAIRPERSON, NARAYANA HEALTH
Dr. Devi Prasad Shetty is the chairman and founder of Narayana Healthand a cardiac surgeon with over34 years of experience. He initiated the concept of micro health insurance schemesin Karnataka, which eventually led to the Karnataka government implementing ‘Yeshasvini’, a micro health insurance scheme forrural farmers. He was a member of the National Innovation Council set up by the Government of India to discuss, analyse and help implement strategies for inclusive innovation in India and prepare a roadmap for innovation 2010-2020. He is the recipient ofa number of awards and honours, most noteworthy being Padma Shri and Padma Bhushanin 2003 and 2012 respectively, conferred by the Government of India.

CHAIRPERSON, MARICO
Mr. Harsh C. Mariwala leads Marico as its chairman. Over the last threedecades, he has transformed a traditional commodity-driven business into a leading consumer products companyin the beauty and wellness space. Marico markets leading brands such as Parachute, Saffola, Mediker, Revive, Setwet, and Livon among others. From a turnover of INR 50 lakhs in 1971, Marico’s products and services in hair care, skin care and healthy foods generated a turnover of about INR 6,300 crores during 2017-18. Today,one out of three Indians is a Marico consumer.

FORMER CHAIRPERSON, ISRO
Dr. Radhakrishnan is former chairman of Indian National Space Organisation (ISRO). Starting his career as an Avionics Engineer in Vikram Sarabhai Space Centre, he held several decisive positions in ISRO in the domains of space launch systems, space applications and space programme management. He is a Fellow of Indian National Academy of Science (FNASc), Fellow of Indian National Academy of Engineering (FNAE); Honorary Life Fellowof The Institution of Engineers, India; Honorary Fellow of The Institution of Electrical and Telecommunication Engineers, India; and Member of International Academy of Astronautics.

PRESIDENT-NASSCOM, AUTHOR
Mr. Kiran Karnik is the Chairperson of Indraprastha Institute of Information Technology, Delhi and HelpAge India, and is also involved with a number of other not-for-profit organizations in the fields of education and development. Earlier, he was Chairperson of Oxfam India (2004-19), and President India Habitat Centre (2006-18). He was a Director on the Board of Reserve Bank of India (2011-15), and now chairs its IT subsidiary, Reserve Bank IT Pvt. Ltd. He is widely recognized for his work in the IT sector, as President NASSCOM from 2001 to 2008. He has had the unique experience of straddling the diverse spheres of government, business and civil society with equal comfort and accomplishment. He was a member of the National Innovation Council set up by the Government of India to discuss, analyse and help implement strategies for inclusive innovation in India and prepare a Roadmap for Innovation 2010-2020. He was awarded the prestigious Padma Shri in 2007.

CHAIRPERSON, BIOCON
Ms. Kiran Mazumdar-Shaw is Chairperson of Biocon since its inception, a first-generation entrepreneur and global business leader with over fourdecades of experience in biotechnology. Fueled by her passion, she started her biotech journey in 1978 from her garage in India. Today, that journey is changing lives for the better across the globe. She is regarded as an unconventional thinker with many firsts to her credit. Under her aegis, Biocon delivers on the promise of making medicines accessible and affordable to millions of patients worldwide.She was also a member of the National Innovation Council set up by the Government of India to discuss, analyse and help implement strategies for inclusive innovation in India and prepare a Roadmap for Innovation 2010-2020. She has received the Padma Shri (1989), Padma Bhushan (2005), Knight of the National Order of the French Legionof Honour (2016), Othmer Gold Medal (2014)and Order of Australia (2020).

FORMER DIRECTOR GENERAL, ICMR
Dr.Nirmal K. Ganguly is Advisor, Translational Health Science and Technology Institute and President, Jawaharlal Institute of Post Graduate Medical Education and Research in India. He is a former Director General of the Indian Council of Medical Research. Dr.Ganguly has published numerous papers and guided doctoral projects. His major research areas are tropical diseases, cardiovascular diseases and diarrhoeal diseases. He is a fellow of several institutions, including the Imperial College Faculty of Medicine and the International Academy of Cardiovascular Sciences, Canada. He has won numerous national and international awards, including the prestigious Padma Bhushanaward from the Government of India.

CO-FOUNDER, SWADES FOUNDATION
Mr. Ronnie Screwvala is a first generation entrepreneur who started his journey by introducing India to multi-channelTV viewing, by launching the first cable TV network in the country. From there, he went on to build a media and entertainment conglomerate spanning television, motion pictures, broadcasting, games and digital, that he listed in 2005 for a market cap of $55 million and that The Walt Disney Company bought in 2012 for an enterprise value of $1.4 billion.Ronnie puts a great deal of emphasis on social welfare. He founded the SwadesFoundation with the plan to impact one million people in rural Maharashtra in five years, with a clear exit/empowerment strategy from those villages, and building a model for others to follow.

FORMER DIRECTOR GENERAL, CSIR
Dr. Shekhar Mande is the Director General of the Council of Scientific and Industrial Research (CSIR) and Secretary of the Department of Scientific and Industrial Research (DSIR), Government of India. He is one of India’s leading experts in DNAfingerprinting and diagnostics. As Director of the National Centre for Cell Science (NCCS), an autonomous Institute of the Department of Biotechnology, Government of India, Dr. Mande oversaw the launch of the Indian Human Microbiome Initiative in the Northeast.His laboratory at NCCS has been involved in research on the structural characterisation of Mycobacterium tuberculosis proteins and the computational analysis of genome-wide protein: protein interactions.In 2005, he was awarded the Shanti Swarup Bhatnagar Prize for Science and Technology –the most prestigious science award in India –in the category of Biological Sciences.

FOUNDER & CH EDITOR, YOURSTORY
Ms. Shradha Sharma is the Founder and Chief Editor of YourStory, which she founded in 2008with the aim of championing the untold stories of entrepreneurship in India.Shradha lives her life in stories and believes thatevery story is uniqueand every story matters. Armed with degrees from St. Stephens College, Delhi and MICA, Ahmedabad, prior to founding YourStory,Shradha worked with Times of India and CNBC TV18. In 2015, she was selected in the list of 500 LinkedIn Influencers across the world. In 2015, she received the L’Oreal Paris Femina Award for Online Influence.

VICE CHAIRPERSON, PIRAMAL GROUP
Dr. Swati Piramal is Vice Chairperson, PiramalGroup, a global business conglomerate with diverse interests in pharma, financial services, healthcare information management, real estate and glass packaging. She is amongst India’s leading scientists and industrialists whose contributions in innovations, new medicines and public health services have touched many lives. Over the last three decades, Dr. Piramal’s work has been focused on the prevention of chronic diseases includingdiabetes, arthritis and heart ailments. Having led a team of scientists, Dr. Piramal has significantly contributed in the field of discovery research. Under her leadership, Piramal Group has been granted over 200 patents in medicine and a library of microbacteria, plants and fungi.

TRUSTEE, ANJANI MASHELKAR FOUNDATION
Sushil Borde heads innovation at Reliance and spearheads strategic initiatives in the Chairman’s office of Reliance Industries Limited, India’s largest private sector firm.
Sushil heads the Anjani Mashelkar Foundation, established by Dr R A Mashelkar, one of India’s legendary innovation leaders. The foundation accelerates disruptive, high-tech, ultra-affordable innovations for transformative impact.
Sushil has also led many other initiatives for Reliance including Mumbai Indians, a cricket franchise of the Indian Premier League (IPL). He was also part of the founding teams of Reliance Foundation and Jio Institute (Reliance’s University in India)
Sushil has spent 7 years in South Africa where he led the Advanced Manufacturing Technology Strategy for the country. Under this programme, he spearheaded an investment agency which deployed public funds in technology programmes to make the South African manufacturing sector globally competitive.
Sushil has also helped MIT, Boston to set up the Fab Foundation in the capacity of its President. The Fab Foundation deploys Fab Labs, a rapid product prototyping set up which allows non-technical people solve local problems through new product development. The mission of the Foundation is ‘Invention as Aid’.
Sushil’s academics include Mechanical Engineering and MBA degrees.